
COVID-19 pandemic has impacted almost every person on this planet and every other business is impacted. The patients enrolled in the clinical trials are unwilling to visit the physician site. Many hospitals and clinical sites have shifted their priorities because of the burden placed by the pandemic. The pandemic has caused disruptions in conducting Clinical Trials forcing Pharmaceutical companies (sponsors) to either clinical trials temporarily or find ways to maintain study continuity. This will bring changes to the way clinical trials are designed and conducted permanently in the future.
Clinicaltrials.gov provides a public database of all the clinical trials registered in the US. Below charts are created based on the latest copy of data downloaded from the CTTI website (https://aact.ctti-clinicaltrials.org/). The report compares the current year numbers of clinical trials submitted, started, completed, and results posted with the previous year.
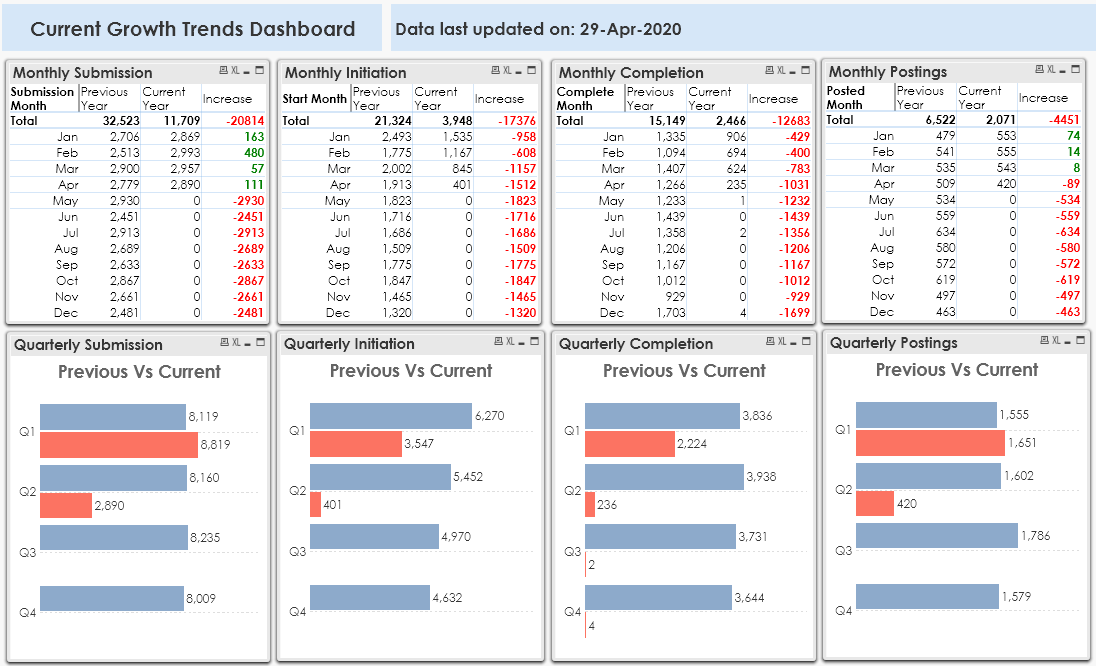
There is an increase in the new studies submitted, however, the March month submission growth is the lowest as compared to Jan and Feb.
To look at the impact of COVID-19, let’s take a look at the number of new studies that started or completed. There is a sharp decline in Feb, Mar, and Apr months. Last month, only 845 new studies were initiated against 2002 studies in March 2019 which is around 60% drop. The current month performed even worse. With just 401 studies being initiated, there is an 80% drop in April, with just 2 days left, and it is unlikely that the numbers would improve before the month-end. Overall Quarter 1 observed an almost 50% decrease in new studies started. We may see a much bigger decline in Quarter 2.
What about the studies that were already in progress? It is difficult to find out what happened to the ongoing studies unless the sponsors disclose themselves. However, we can get a sense of it by looking at the number of studies completed in the last few months. The studies that would have been very close to completion with only a few patient visits pending, should have appeared in the number of completed studies for March and April months. The studies completed figures have dropped by around 42% in the First Quarter. Mar and Apr month completed studies figures have dropped by 55% and 81% respectively. Any delay in completing the trials can bring complexities in estimating the accuracy of the data and efficacy of the investigational new drug.
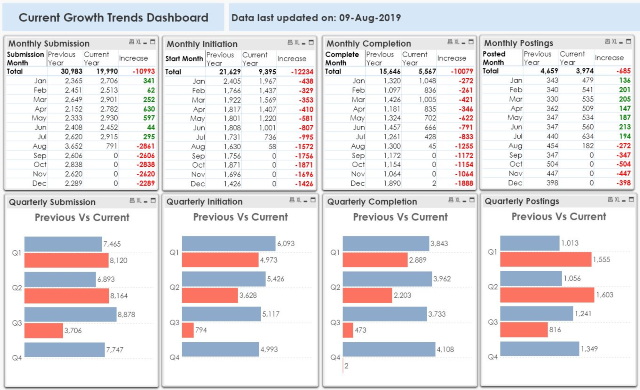
The above chart shows how it looked like in Aug 2019. The study initiation and completion figures were also somewhat lower when compared to the numbers in the previous year for the same month. However, COVID-19 has made it worse now.
Disruptions in recruiting new patients and then screening them might have also impacted the new studies to start. If the study sites do not find sufficient patients, the study cannot begin. As of today, there are 7,652 sponsors currently recruiting patients for 51,817 studies in 169 countries.
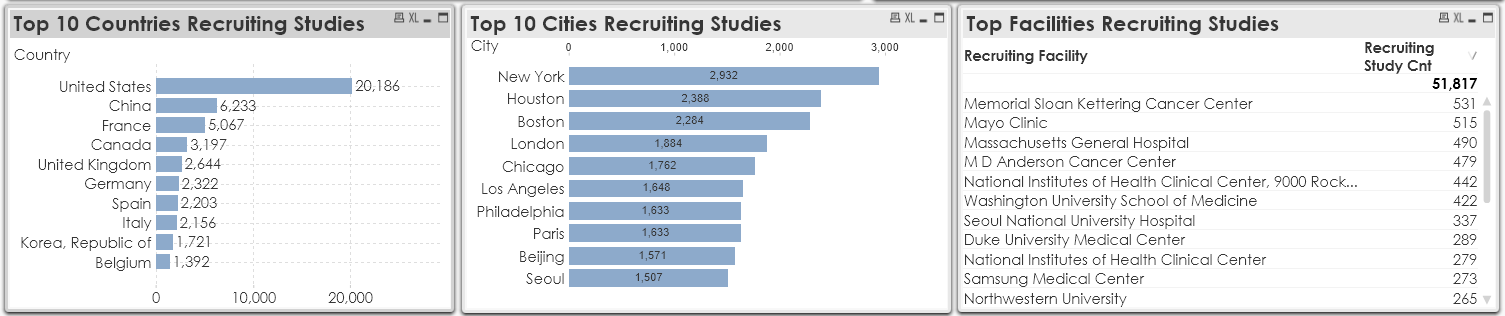
The above chart shows the top 10 countries and cities currently recruiting. All the top recruiting countries are also the worst affected countries from COVID-19. The top recruiting city New York has become the epicenter of the pandemic with almost 17,671 fatal coronavirus cases. Most of the top recruiting facilities are bigger hospitals that are overwhelmed by the burden placed by the pandemic. Obviously, clinical trials are not the priority at this time for those facilities. It is possible that sponsors and recruiting agencies can continue to find new patients through social media and other channels that may not require the patient to make a physical visit to the clinical site. However, the patients have to be screened first to check if they are eligible to participate or not.
In many cases, screening requires laboratory tests to be conducted for which patients are required to visit the site or a partner laboratory.
We can see that the COVID-19 has effected around 60% of studies to get started. The studies that were not initiated will impact the overall ecosystem. The resources that would have been engaged to conduct those trials are also affected which includes CRO (Contract Research Organizations), physicians, and of course patients. The biggest loss is that the patients will be deprived of the investigational drugs to cure their sufferings. The patients are anxiously waiting for the clinical trials to begin soon. The study delays would mean the delay in bringing the new drug to the market making the patients wait longer.
There are economic impacts associated with the delay in clinical studies. Many contractual businesses that provide their services to the study sponsors might have got impacted. Contract terminations and delays in payments may force many to shut down or reduce their workforce. Pharma companies rely heavily on building and maintaining a strong pipeline of patents for continuous future growth projections. Smaller pharma or biotech companies rely heavily on a few promising drugs, and any risk posed to the drug development process can have a devastating effect on their businesses.
COVID-19 has caused some positive developments in the clinical research industry. The challenges posed by the pandemic has encouraged sponsors and regulators to adopt technological advances to improve the entire clinical development lifecycle. Sponsor companies are finding ways to reduce the burden on patients by reducing patient visits using technologies used in telemedicine (virtual nurse and physician consultations), home nursing, direct-to-patient delivery of drugs. Companies are acting to modify or create clinical protocols that are more patient-centric eliminating any unnecessary patient visits and procedures. Sponsors are partnering with technology companies to set up the infrastructure to conduct trials in a virtual setting using mobile devices completely or in a hybrid format.
It is yet to be seen how the industry would recover from the crisis, however, it is clear that the future trials will be designed and conducted in a way that is more patient-centric and virtual.
—————————————————————————————————————————————-
Disclaimer: The author has no affiliation with any government or political party or organization, and the views expressed are author’s personal views and do not represent the opinion of any entity whatsoever with which the author has been, is now or will be affiliated. The author does not accept any responsibility or liability for any direct, indirect, or consequential loss or damage resulting from any such irregularity, inaccuracy, or use of the information.











